What Can Be Done to Mitigate Antimicrobial Resistance's Threat to Global Health?
Humanity is entering a dangerous era in which common infections that were once easily cured are regaining their power to kill. Antimicrobial resistance (AMR) – the process by which microorganisms evolve to resist the effects of antimicrobial medicines – is rendering previously effective treatments ineffective, and eroding the foundations of modern medicine. Driven largely by the misuse and overuse of antibiotics in human healthcare, animal medicine, agriculture, and beyond, resistant pathogens are emerging and spreading to threaten both human and animal health worldwide. The scope of the problem is vast: without effective antimicrobials, routine surgeries, cancer treatments, and even the management of minor injuries become high-risk. With over 1.2 million deaths linked to AMR in 2019 and projections of up to 10 million annually by 2050, AMR is recognised as one of the most significant and pressing threats to global health security facing the 21st century, and it cannot be resolved by any one organisation or nation acting alone. If there is no immediate, harmonised global action, the world risks returning to an era where common infections once again take millions of lives.
The impacts of AMR are far-reaching, spanning health, economic, and societal domains. Directly, it drives higher morbidity and mortality, strips away effective treatment options for common diseases, compromises the safety of medical procedures, and forces dependence on costly and often more toxic last-resort drugs. Infections with resistant microorganisms lead to longer illnesses, prolonged hospital stays, and higher healthcare costs. AMR also presents significant cross-border and One Health risks, spreading quickly through travel, trade, migration, and food supply chains and moving between humans, animals, and the environment. Food production plays an especially critical role in this transmission, as resistant bacteria from livestock and food products can be passed to humans. Economically, AMR drains productivity by causing sickness in both people and animals, undermines food security, and imposes heavy financial burdens – such as the €1500 million annual cost estimate for the European Union. These challenges are fuelled by widespread misuse and overuse of antibiotics in medicine and agriculture, inadequate infection prevention and sanitation, poor regulation, circulation of substandard or counterfeit drugs, and a stagnating drug development pipeline with no major new class of antibiotics discovered since 1987.
Recognising the urgent need for coordinated intervention, the World Health Assembly in May 2015 adopted a Global Action Plan on antimicrobial resistance built around five strategic objectives. First, improving awareness and understanding of AMR through targeted communication, education, and training ensures that the issue becomes embedded in professional development and school curricula. Second, strengthening the evidence base through surveillance and research is critical, yet hampered by the absence of internationally agreed standards for data collection and reporting. Third, reducing infection incidence via effective sanitation, hygiene, and infection prevention – alongside widespread vaccination of both humans and animals – can diminish the need for antimicrobials and curb inappropriate use for viral infections. Fourth, optimising the use of antimicrobial medicines in human and animal health requires better diagnostics, evidence-based prescribing, stricter regulation of sales, and robust monitoring, particularly in low-income countries. Finally, economically incentivising research into new medicines, diagnostics, and interventions and ensuring affordable access to new medicines is vital. This World Health Organisation (WHO) framework calls for cross-sectoral coordination spanning human and veterinary medicine, agriculture, finance, and environmental management, while addressing the disparities in national resources and the economic challenges that have inhibited research and innovation.
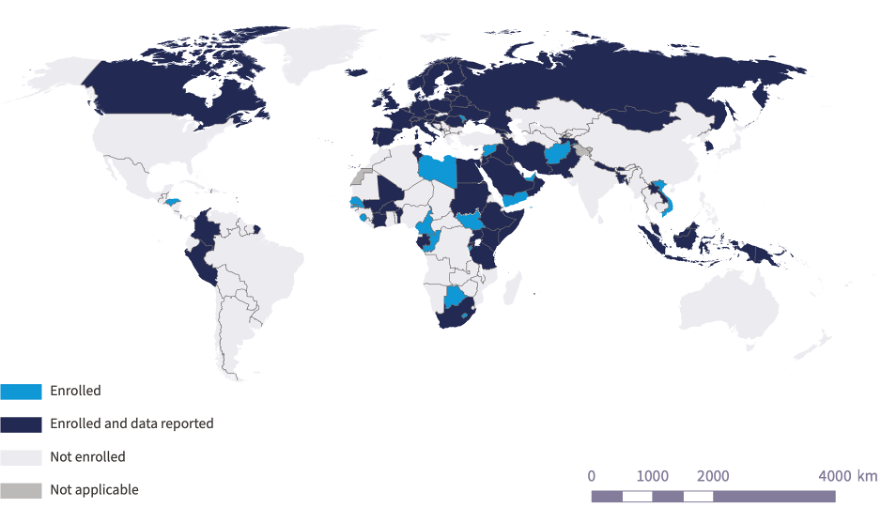
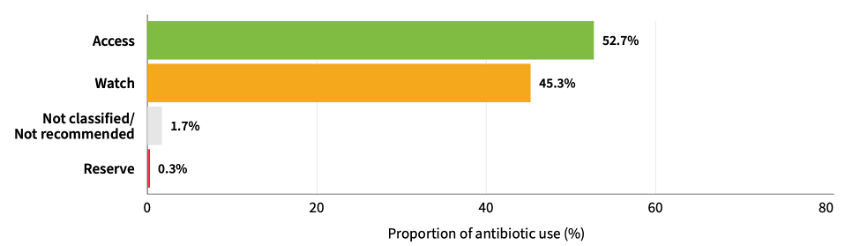
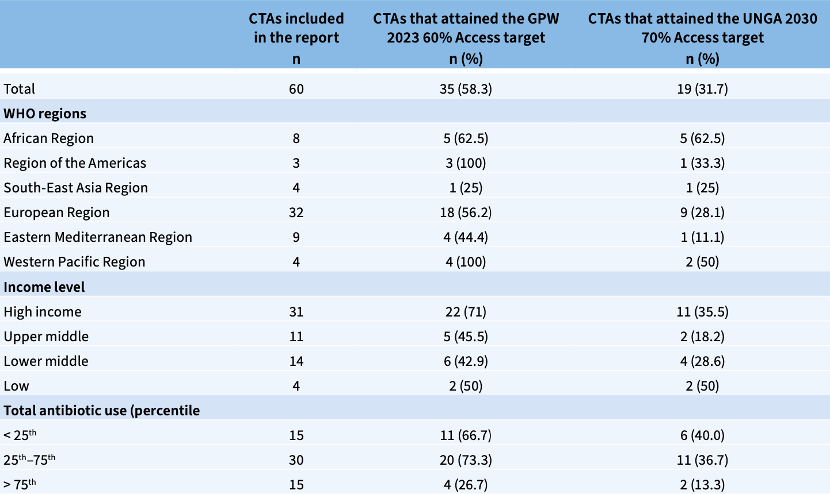
Current surveillance and progress on antimicrobial use (AMU) and antimicrobial resistance have shown measurable but uneven advances. The WHO Monitoring and Evaluation (M&E) Framework, established in 2019, was designed to track the implementation and impact of the Global Action Plan (GAP) on AMR, assessing both processes and outcomes through defined indicators. The framework does not set global targets due to wide variability among countries, but it enables structured data collection and evaluation through systems like the Tripartite AMR Country Self-Assessment Survey (TrACSS), the Global Antimicrobial Resistance Surveillance System for Antimicrobial Use (GLASS-AMU), and other country- and regional-level initiatives. By 2022, GLASS-AMU data from 60 countries, territories, and areas (CTAs) covering 1.74 billion people reported a total of 16.6 billion defined daily doses (DDDs) of antibiotics. Of these, as classified by the AWaRe categorisation system for the guidance of appropriate antibiotic use, 52.7% were Access antibiotics – representing an overall failure to meet the WHO General Programme of Work (GPW) 2023 Access target of 60% (though 58% of CTAs met this target nationally). 45.3% of the total DDDs were Watch antibiotics, with Reserve antibiotics accounting for only 0.3% and eight CTAs in low- and middle-income countries (LMICs) reporting no Reserve antibiotic use at all, reflecting issues of availability and affordability. Participation in GLASS-AMU has steadily increased since its inception, yet it remains skewed toward high-income countries, with the highest relative participation in the Eastern Mediterranean region – which also reported highest overall levels of antibiotic consumption, alongside Southeast Asia – and lowest participation in the Americas. Conclusively, the data reveals significant variations in surveillance and antibiotic use across regions, countries, and antibiotic classes, emphasising the need for sustained monitoring, targeted capacity building, and global interventions to ensure equitable access to essential antibiotics while minimising unnecessary use of Watch and Reserve drugs.
Mitigating the threat of AMR will require a comprehensive set of strategies that combine stronger policy, enhanced surveillance, improved infection prevention, optimised antibiotic use, and renewed investment in innovation. At a policy level, governments must enforce regulations that limit inappropriate access to antibiotics, particularly where over-the-counter and online sales continue to drive unnecessary consumption, while expanding participation in surveillance platforms such as GLASS-AMU to address current gaps in data quality and the under-representation of LMICS. Strengthening surveillance also demands standardised, internationally agreed upon methods of data collection and reporting, alongside a One Health approach that integrates human, animal, and environmental monitoring to better trace the emergence and spread of resistant pathogens. Preventing infections at their source remains essential: investment in sanitation, hygiene, and WASH infrastructure, the expansion of immunisation programmes for both humans and animals, and rigorous hospital infection-control practices can significantly reduce the need for antibiotic use. Stewardship measures must ensure that antibiotics are used responsibly through national implementation of AWaRe prescribing targets, wider availability of rapid and affordable diagnostics to curb empiric prescribing, and sustained public and professional education on the rational use of these medicines. Finally, long-term progress requires international collaboration, including economic pipelines, develop new diagnostics and vaccines, and ensure that all countries – especially those with limited resources – have equitable access to existing essential antibiotics as well as any new products developed to combat multidrug-resistant infections.
AMR is a transnational, multisectoral crisis that is steadily eroding the foundation of modern medicine as the rapid evolution of resistant microorganisms outpaces the development of new treatments. Driven by the widespread overuse of antibiotics in human health care and agriculture, this resistance now threatens to push the world further into a post-antibiotic era in which once-manageable infections again become life-threatening and routine medical procedures carry high risks. Preventing this outcome requires more than isolated national efforts: the 'tragedy of the commons' nature of AMR demands immediate, coordinated global action grounded in a One Health approach that unites human, environmental, and animal health. Only through long-term political commitment and genuine international cooperation can antimicrobial effectiveness be preserved for future generations.
Image courtesy of Wilfredor via Wikimedia ©2017. Some rights reserved.
The views and opinions expressed in this article are those of the author and do not necessarily reflect those of the wider St Andrews Foreign Affairs Review team.